Electrochemical reactions are used by most mobile devices such as laptop computers and mobile phones to release energy from batteries. Liquid hydrocarbons (fuels such as gasoline, diesel and kerosene) are today the densest way known to economically store and transport chemical energy at a large scale (1 kg of diesel fuel burns with the oxygen contained in ≈15 kg of air). Chemical reactions are used by organisms to derive energy from food and by automobiles to derive energy from gasoline. Nuclear reactions take place in stars and nuclear power plants, both of which derive energy from the binding energy of nuclei. In order of the typical magnitude of the energy released, these types of reactions are: nuclear, chemical, electrochemical, and electrical. There are different types of energy stored in materials, and it takes a particular type of reaction to release each type of energy. A pressure gradient describes the potential to perform work on the surroundings by converting internal energy to work until equilibrium is reached. Likewise, the energy required to compress a gas to a certain volume may be determined by multiplying the difference between the gas pressure and the external pressure by the change in volume. For example, the energy density of a magnetic field may be expressed as and behaves like a physical pressure. 
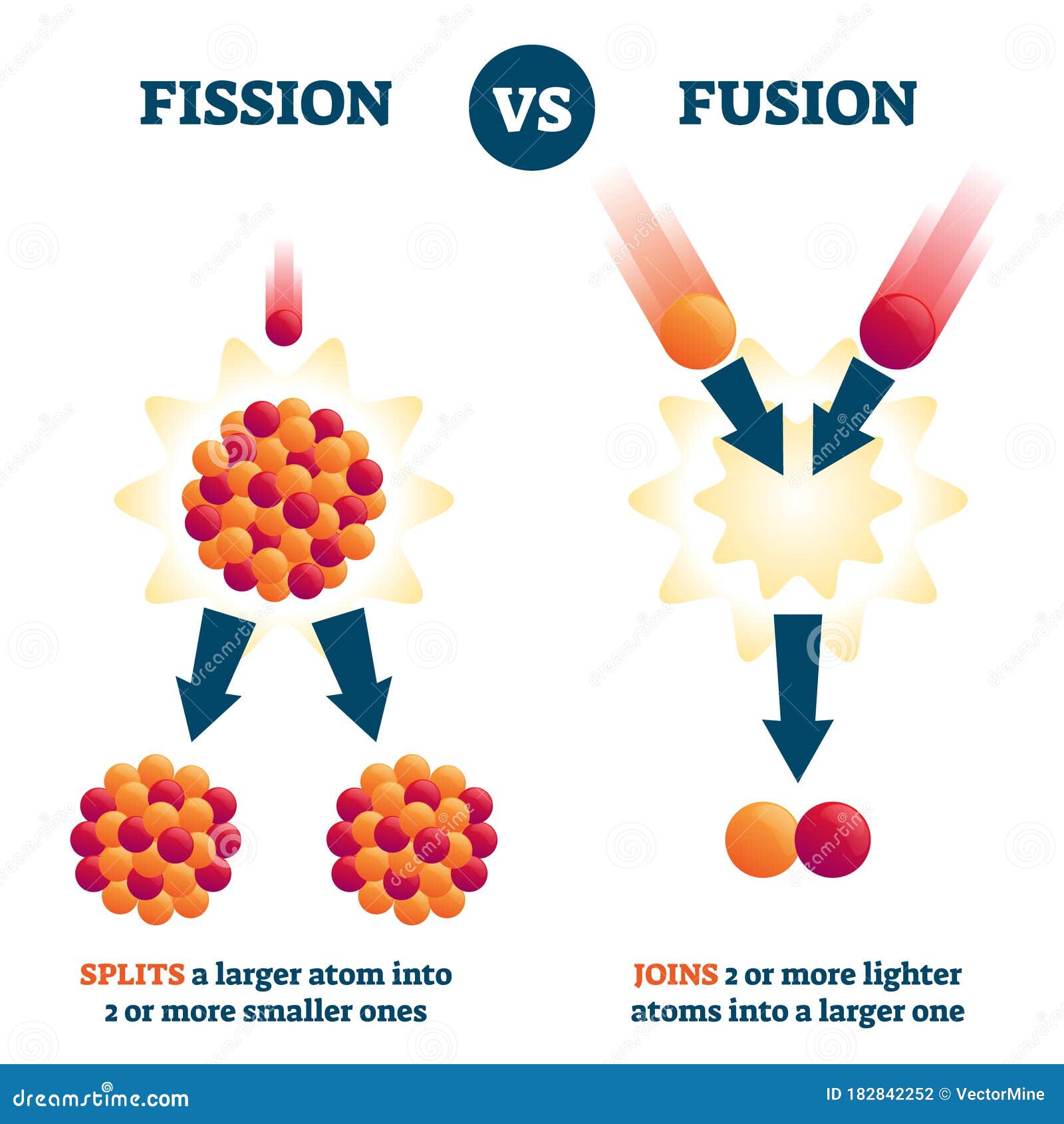
In cosmological and other general relativistic contexts, however, the energy densities considered are those that correspond to the elements of the stress-energy tensor and therefore do include mass energy as well as energy densities associated with pressure.Įnergy per unit volume has the same physical units as pressure and in many situations is synonymous. Often only the useful or extractable energy is measured, which is to say that inaccessible energy (such as rest mass energy) is ignored. It is sometimes confused with energy per unit mass which is properly called specific energy or gravimetric energy density. In physics, energy density is the amount of energy stored in a given system or region of space per unit volume. So the ratio of mass destroyed in fusion of hydrogen is 8 times greater than the mass destroyed during the fission of uranium, which is a way of saying that fusion produces 8 times more energy per mass than fission does.L − 1 M T − 2 So that's 6.4 times 10 to the 14 joules divided by 8 times 10 to the 13 joules and this gives us a factor of 8. And so that means they cancel and we have the ΔM H over ΔM u is gonna be the energy released during the fusion of hydrogen over c squared divided by the energy released during the fission of uranium over c squared the c squared's cancel and so we can divide these energies directly. So we have these two ratios divided by each other and the mass of hydrogen is the same as the original mass of uranium because in this table, it's 1 kilogram in each case. And so this line here tells us that when 1 kilogram of uranium is fissioned, it releases 8 times 10 to the 13 joules of energy. And then in part (c), it asks us to compare the ratio of of hydrogen that has turned into energy during fusion versus the ratio of uranium turned into energy during fission. And we divide that by the original mass of 1 kilogram, which we're told here, and that is 0.0071 so 0.7 percent has turned into energy. And the fraction of hydrogen that has turned into energy is gonna be this amount of mass that's lost, which written as kilograms is 0.00712. So the amount of mass of hydrogen that has turned into energy is 6.4 times 10 to the 14 joules of energy divided by speed of light squared which is 7.1 grams. And so we'll divide both sides by c squared and we get the mass is energy divided by c squared. And so the question is, how much mass disappears and turns into energy? So the energy released is gonna be the amount of mass that disappears times c squared. When 1 kilogram of hydrogen is fused into helium, we're told that 6.4 times 10 to the 14 joules of energy are released. This is College Physics Answers with Shaun Dychko.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
